The Carbon Dioxide Story
The carbon cycle is important to understand because it explains the ways that 1) carbon dioxide is produced and consumed through natural processes and 2) humans are able to rapidly alter the cycle in a very short span of years. Carbon dioxide (CO2), the major greenhouse gas of concern, provides the predominant way that carbon is cycled throughout natural reservoirs.
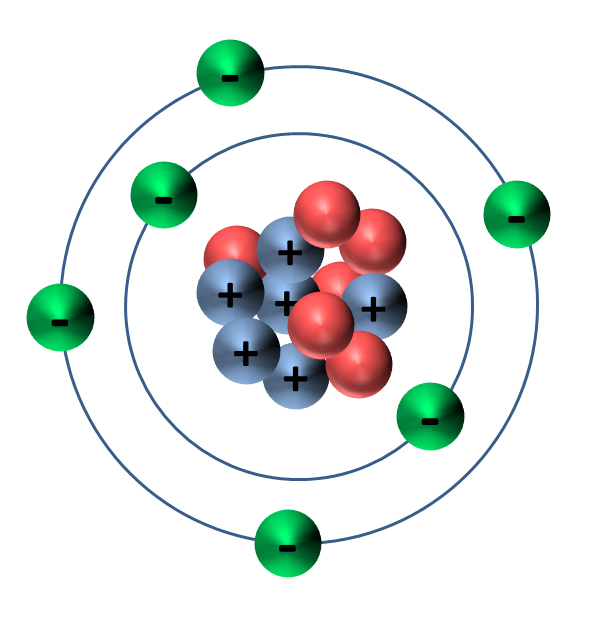
Carbon
Pure carbon is very rare. It is found in nature only as the minerals, graphite and diamond. Most carbon is bonded to other elements to form compounds. Carbon, “the building block of life,” combines with hydrogen and oxygen to form the basic compounds that make up living things. We (as well as all of the other plants and animals on earth) are made of carbon, as much as 50% of our dry weight. It is the element that all organic substances contain – from fossil fuels to DNA. Hydrocarbons – such as coal, oil (also known as petroleum, and natural gas) – are compounds made of hydrogen and carbon. Carbon is also found in calcite (CaCo3), a mineral in sedimentary and metamorphic rocks, such as limestone and marble.
Carbon is found on Earth in three large reservoirs as 1) carbon dioxide in the atmosphere, 2) dissolved carbon dioxide in the oceans, and 3) coal, oil, natural gas, and calcium carbonate rock underground. Carbon cycles through all four of Earth’s spheres (the atmosphere, hydrosphere, geosphere, and biosphere) on different time scales. The cycle is known as the carbon cycle. Carbon is primarily cycled in the form of carbon dioxide (CO2) between the natural carbon reservoirs.
The Carbon Cycle
Carbon dioxide (CO2) only makes up 0.040% of the total atmosphere. However, it is the major greenhouse gas that contributes to global warming for two reasons--it has a relatively long lifetime in the atmosphere (approximately 100 years) and it is a strong absorber of infrared radiation.
Carbon dioxide is added to the atmosphere by human activities. When hydrocarbon fuels (i.e. wood, coal, natural gas, gasoline, and oil) are burned, carbon dioxide is released. During combustion or burning, carbon from fossil fuels combine with oxygen in the air to form carbon dioxide and water vapor.
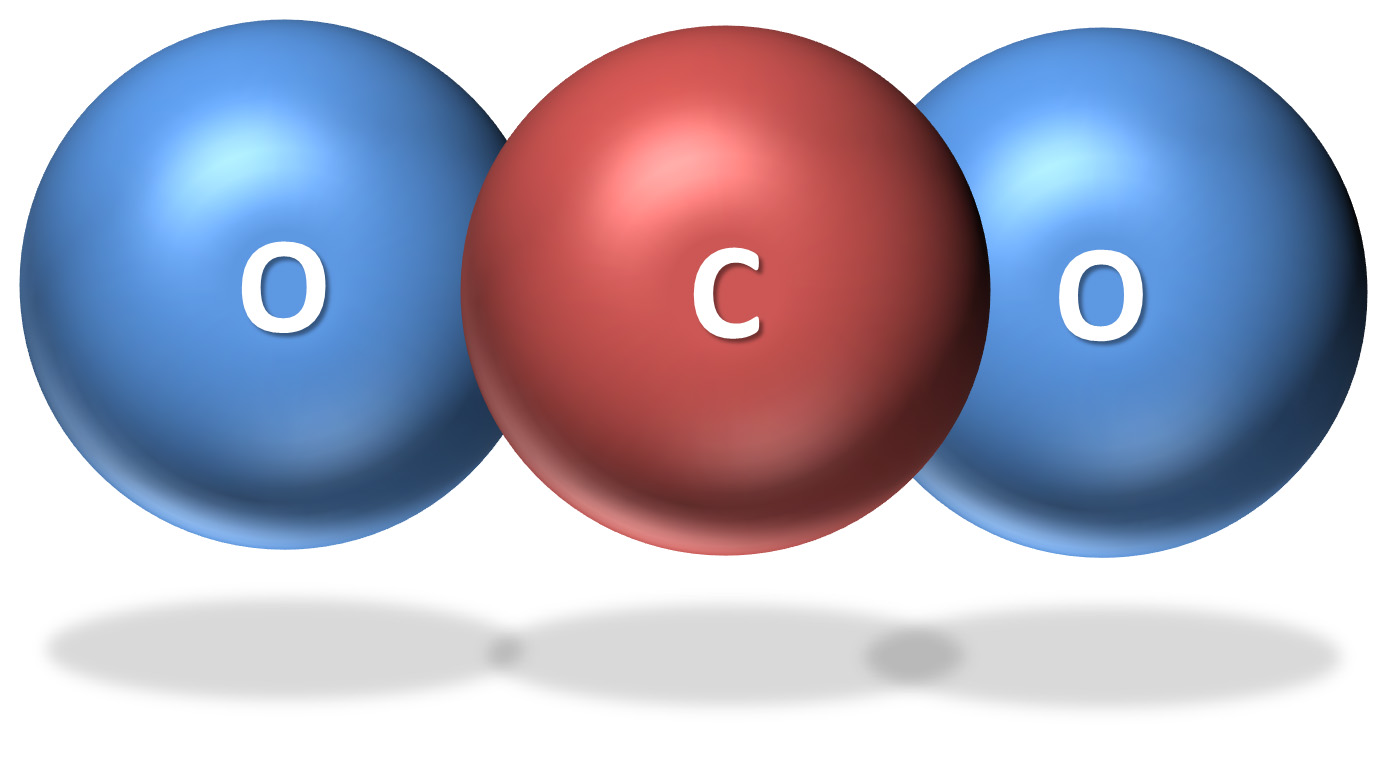 Carbon dioxide consists of one atom of carbon and two atoms of oxygen, which are held together by covalent bonds (or the sharing of electrons). When agitated by infrared radiation, the CO2 molecule vibrates in all three directions and absorbs heat. It is then able to re-radiate or emit heat in all directions, including back toward Earth. Hence, carbon dioxide absorbs infrared radiation emitted from Earth’s surface before re-emitting the same infrared radiation as was absorbed.
Carbon dioxide consists of one atom of carbon and two atoms of oxygen, which are held together by covalent bonds (or the sharing of electrons). When agitated by infrared radiation, the CO2 molecule vibrates in all three directions and absorbs heat. It is then able to re-radiate or emit heat in all directions, including back toward Earth. Hence, carbon dioxide absorbs infrared radiation emitted from Earth’s surface before re-emitting the same infrared radiation as was absorbed.
Watch Episode 1: Global Warming, It's All About Carbon.
NPR's Robert Krulwich and Odd Todd, in partnership with Wild Chronicles, present an animated cartoon series on the atom at the heart of global warming: carbon.
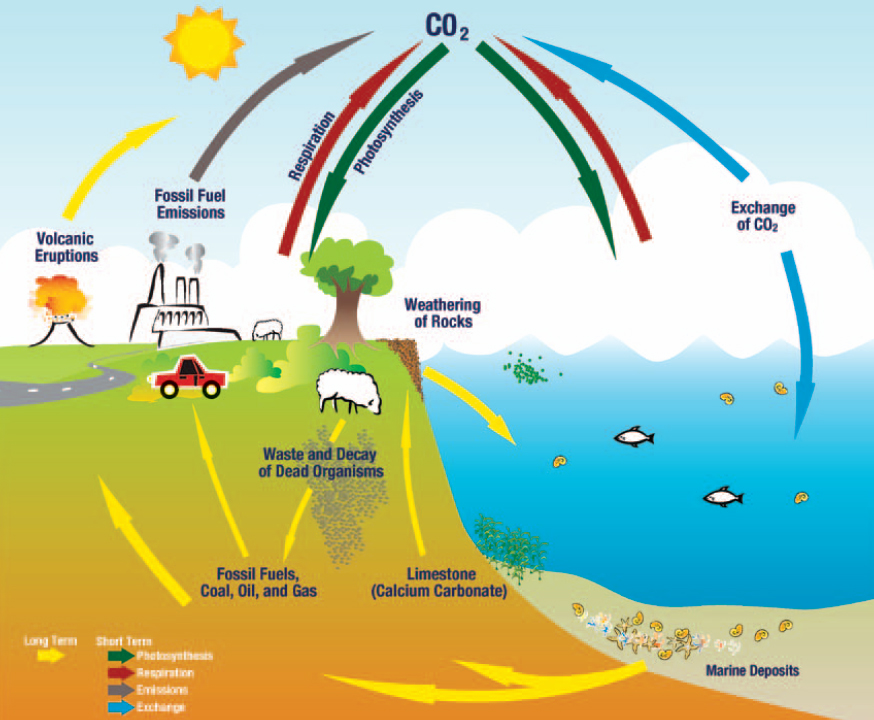
Carbon dioxide constantly moves into and out of the atmosphere through several major pathways. Over short time scales, the processes of photosynthesis, respiration, organic decomposition (decay), and combustion (burning of organic material) increase or decrease the concentration of atmospheric CO2. Carbon dioxide is also exchanged between the atmosphere and oceans by gas exchange over short time scales. Each year, approximately one-fifth of the carbon (in the form of CO2) in the atmosphere is cycled in and out.
Over longer time scales, the concentration of atmospheric CO2 is changed by the formation of fossil fuels, weathering of rocks, and volcanic eruptions. The following sections describe these short- and long-term processes.